
Have our bromine here which we have three Our carbon has a plus one formal charge and we are also gonna So we have a carbon that isīonded to three methyl groups and this is a plainer carbocation so I'm trying to show that. Off on to the bromine to form our bromide anion and we are gonna formĪ carbocation as well. So down here I have tert butyl bromide and for an SN1 mechanism the first step here wouldīe loss of a leaving group so these electrons come So an SN1 mechanism is favored by a polar protic solvent and let's look at why. So these polar protic solvents favor an SN1 mechanism. Here is our hydrogenĪnd here is our oxygen. Which again has a hydrogen directly connected toĪn electronegative atom and oxygen and finally acetic acid which Water is an example ofĪ polar protic solvent. Hydrogen directly connected to an electronegativeĪtom which is oxygen. For example if you look at water here, you can see we have a A polar protic solvent is a solvent that has at least one hydrogen connected to an electronegative atom. Although only a bookkeeping device, formal charges often give clues about chemical reactivity, so it’s helpful to be able to identify and calculate them correctly.A solvent can have an effect on an SN1 or an SN2 mechanism. The number of electrons in the bonded atom, in turn, is equal to half the number of bonding electrons plus the nonbonding, lone-pair electrons.Ī summary of commonly encountered formal charges and the bonding situations in which they occur is given in Table 1. To express the calculations in a general way, the formal charge on an atom is equal to the number of valence electrons in a neutral, isolated atom minus the number of electrons owned by that bonded atom in a molecule. Atomic oxygen has six valence electrons, but the oxygen in dimethyl sulfoxide has seven-one in the O- S bond and two in each of three lone pairs. 
A similar calculation for the oxygen atom shows that it has formally gained an electron and has a negative charge. Thus, the sulfur atom has formally lost an electron and therefore has a positive charge. Atomic sulfur has six valence electrons, but the dimethyl sulfoxide sulfur owns only five-one in each of the two S- C single bonds, one in the S- O single bond, and two in a lone pair. 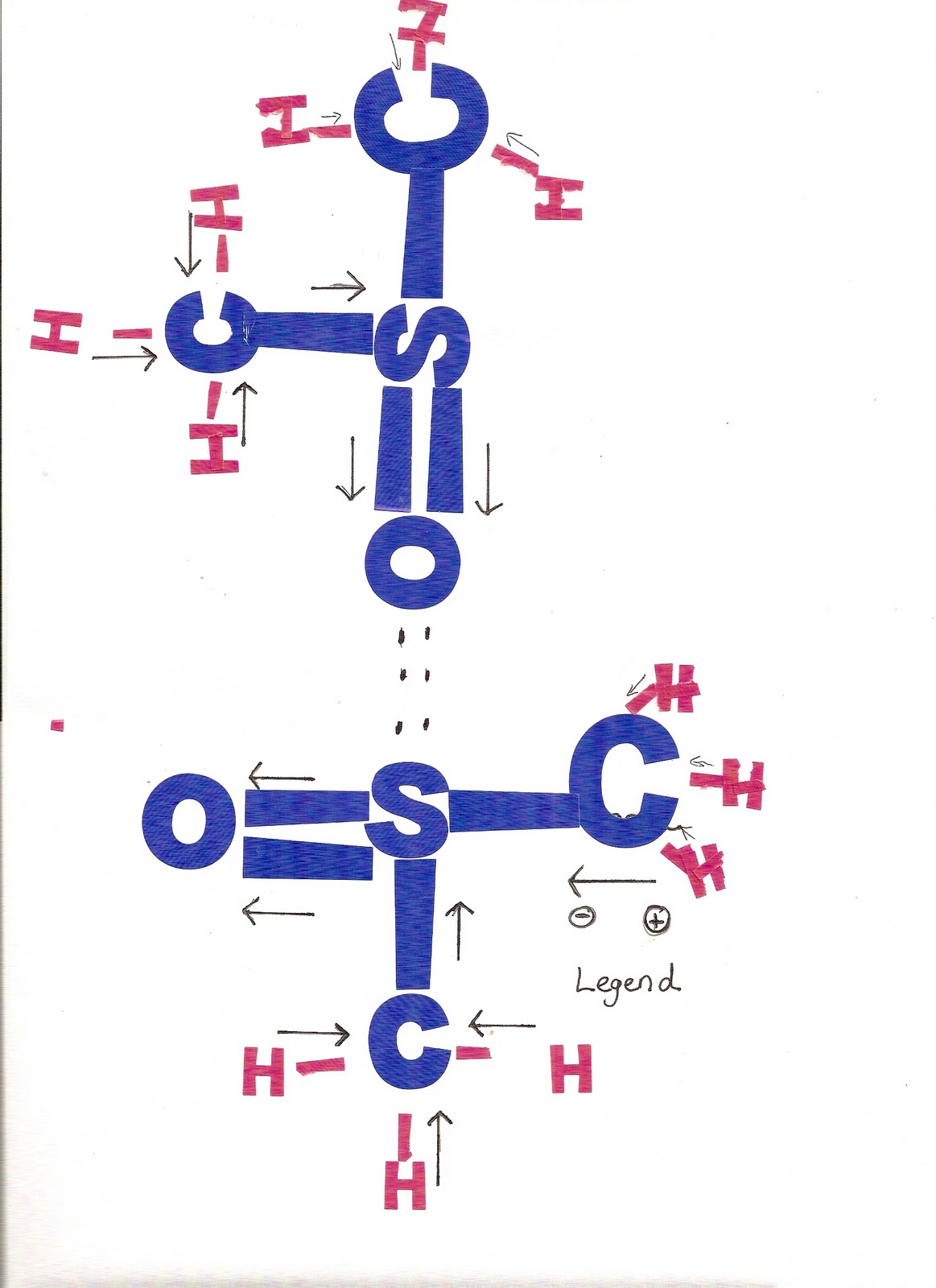
The situation is different in dimethyl sulfoxide. Thus, the nitrogen atom in ammonia has no formal charge. Atomic nitrogen has five valence electrons, and the ammonia nitrogen also has five-one in each of three shared N- H bonds plus two in the lone pair. The same is true for the nitrogen atom in ammonia, which has three covalent N- H bonds and two nonbonding electrons (a lone pair). Because a neutral, isolated carbon atom has four valence electrons, and because the carbon atom in methane still owns four, the methane carbon atom is neutral and has no formal charge. In methane, for instance, the carbon atom owns one electron in each of the four C - H bonds. Although the bonding electrons are shared by both atoms, each atom can still be considered to “own” one electron for bookkeeping purposes. Instead, they’re a device for electron “bookkeeping” and can be thought of in the following way: a typical covalent bond is formed when each atom donates one electron. 
Note that an electrostatic potential map of dimethyl sulfoxide shows the oxygen as negative (red) and the sulfur as relatively positive (blue), in accordance with the formal charges.įormal charges, as the name suggests, are a formalism and don’t imply the presence of actual ionic charges in a molecule. The oxygen atom, by contrast, has one bond rather than the usual two and has a formal negative charge. The sulfur atom in dimethyl sulfoxide has three bonds rather than the usual two and has a formal positive charge. Look at dimethyl sulfoxide (CH 3SOCH 3), for instance, a solvent commonly used for preserving biological cell lines at low temperature. Closely related to the ideas of bond polarity and dipole moment is the concept of assigning formal charges to specific atoms within a molecule, particularly atoms that have an apparently “abnormal” number of bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
